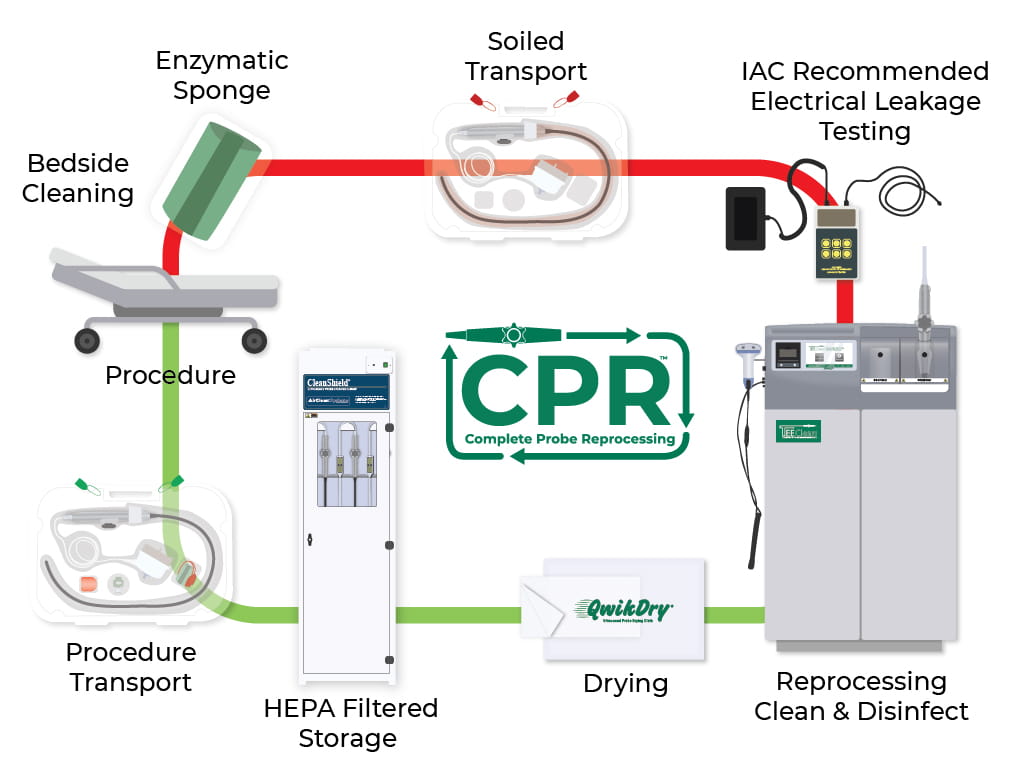
For years CS Medical has built our TEE probe reprocessing model around our ten-step program, but as technology has advanced, this model has slowly become outdated. We have made enormous strides in automation, a science-backed method to reduce error and guarantee uniform, superior results. As a result, we're pleased to introduce the latest model in TEE probe reprocessing: Complete Probe Reprocessing.
With Complete Probe Reprocessing, there are now just seven steps to keep track of when reprocessing a TEE ultrasound probe:
-
Point of Use Cleaning
Immediately upon removing the TEE probe from the patient, the probe must be bedside cleaned (also known as pre-cleaning or point of use cleaning). A pre-soaked sponge, like the TEEZyme® Enzymatic Sponge, is the best tool to remove organic and inorganic soil from the insertion tube and to prevent any remaining soil from drying on the probe. This step is required for the successful reprocessing of TEE probes.
-
Soiled TEE Probe Transportation
The soiled TEE probe must next be transported to the proper location for reprocessing. The best solution for this step will keep the probe moist, protect the probe from damage, protect staff and patients from exposure, and clearly label the probe as dirty. The TPorter® TEE Ultrasound Probe Transportation Case with the was carefully designed to provide standardized care and handling of soiled TEE probes during transportation.
Electrical Leakage Testing
Electrical leakage testing must be performed between every use of a TEE probe; this ensures the safety of patients and staff, preventing a damaged probe from being used on a patient. Consequences from using a probe that has an electrical problem could range from inconclusive testing to even death of a patient; electrical leakage testing must never be skipped and is required for all IAC accredited facilities. CS Medical offers the ULT-2020 ultrasound leakage tester and respective adapters that are specific to each TEE probe. No matter which TEE probes your facility uses, we've got you covered.
Cleaning and High-Level Disinfection
What once took up a full four steps has now been whittled down to one simple step: TEEClean® Automated TEE Probe Cleaner Disinfector provides both manufacturers recommended cleaning and high-level disinfection of soiled TEE probes in the same FDA-cleared device. By using the TEEClean to automate the cleaning and disinfecting steps, you save your facility time, money, and effort. This science-based solution provides consistent, superior results every time.
TEE Probe Drying
Once a TEE probe has been fully cleaned, disinfected, and rinsed in the TEEClean, it must be thoroughly dried to prevent recontamination and the growth of bacteria. QwikDry® Ultrasound Drying Cloths are gamma-irradiated, meaning they won't introduce bacteria to the probe. And QwikDry's super absorbent matrix and ultra-smooth surface will help ensure the TEE probe's insertion tube will be absolutely dry and ready for storage.
TEE Probe Storage
Once a probe has been disinfected and dried, it is vital that the method of storage prevents recontamination and protects the probe from any damage. The CleanShield® TEE Ultrasound Probe Storage Cabinet is one excellent option available. Not only will the positive pressure HEPA filtered clean air minimize the chance of airborne contaminants entering into the cabinet, but the CleanShield also meets the most current Joint Commission standards for care.
Clean TEE Probe Transportation
When the TEE probe is ready to be used again, transportation to the procedure room must be a secure process that also prevents contamination of the probe. Employing the TPorter® TEE Ultrasound Probe Procedure Case with the is a perfect way to protect the probe and also ensure that you have everything you need to perform the TEE procedure. The TPorter conveniently includes spaces for each item required for a typical TEE procedure and even had spaces for "clean" and "dirty" tags, to properly label the TEE probe for all to see.
At CS Medical, we are always excited to come alongside you and help make your job a little simpler so you can focus on the most important part of what all of us in the healthcare industry want: to help people get and stay healthy.


