In ultrasound exam rooms, the way the space is designed impacts more than just convenience; it directly affects infection prevention. When staff need to turn over rooms quickly, the layout plays a crucial role in making it easy to follow the correct reprocessing workflow. If the only accessible water source is a handwashing sink, staff may end up using it for probe cleaning, unintentionally introducing infection risk into the process.
This is why infection prevention stakeholders should be involved early — before equipment is purchased or placed, and before plumbing is discussed. The goal isn’t to make rooms more complicated; it’s to make the safest process the easiest process. A dedicated, consistent reprocessing workflow should not compete with hand hygiene and should not create splash-zone contamination risk.
A handwashing sink is intended only to support hand hygiene, period. Multiple standards and best practice guidelines emphasize that hand-hygiene sinks should be separate from cleaning and reprocessing sinks. The moment you mix purposes, you increase the risk of cross-contamination and create a splashing or aerosol problem.
Why does this matter specifically for ultrasound probe cleaning?
- • Splash-zone contamination is real. Water striking drains or faucets can spread contaminated droplets to nearby surfaces and items. Splashes pose an exposure concern, as evidenced in the literature. A study by Cori Ofstead and team demonstrates that droplets can travel as far as 7 feet from the sink where medical devices are cleaned.
- • Survey and compliance expectations recognize a “splash zone.” Regulatory survey guidance commonly uses 3 feet as the distance at which splashing water can contaminate nearby clean activities and items, including medication prep and clean supplies.
- • Reprocessing guidance separates handwashing from reprocessing sinks. For example, infection prevention standards for medical device reprocessing in any location require a handwashing sink separate from the reprocessing sink.
- • AORN guidance for instrument cleaning specifically warns against using hand-hygiene sinks. AORN’s instrument cleaning guidance states not to clean/decontaminate instruments in sinks where hand hygiene is performed.
- • AAMI standards also reinforce sink separation in device processing areas. ANSI/AAMI ST58:2024 (chemical sterilization/high-level disinfection) includes requirements consistent with handwashing sinks being designated and separate from sinks used for processing medical devices.
- • CDC environmental cleaning guidance also supports the use of dedicated sinks for reprocessing tasks. CDC best-practice guidance for environmental cleaning programs notes that utility sinks/drains should be available and distinguishes them from sinks used for hand hygiene.
If staff are rinsing or cleaning probes in (or right next to) the handwashing sink, you’ve created (1) cross-traffic between dirty and clean tasks, and (2) splash-zone opportunities that can contaminate nearby clean storage, counters, supplies, and even hands after handwashing.
Ethos® Automated Ultrasound Probe Cleaner Disinfector: Designed for the Room You Already Have
Ethos is designed to automate probe cleaning and high-level disinfection in a single device, incorporating a filtered water rinse as part of a workflow aligned with AAMI ST108 recommendations. This workflow doesn’t depend on a handwashing station for anything other than its availability for handwashing.
- • This matters to infection preventionists because it helps the room support:
- •Dedicated reprocessing behavior and workflow
- • Reduced handling/variation
- • A reprocessing flow that doesn’t compete with hand hygiene
In many ultrasound exam rooms, space is limited, and staff often assume that adding automated probe reprocessing equipment will require significant renovation. In reality, systems such as Ethos can often be positioned beside existing cabinetry and plumbing with minimal disruption to the room layout. In fact, Ethos can easily fit into any exam room and tie into the plumbing already under the handwashing sink, avoiding costly renovations.
In the below example, the system fits naturally alongside the counter and sink area, allowing connection to existing plumbing while keeping the exam space fully functional for patient care. This configuration also reinforces an important infection prevention principle: the handwashing sink remains dedicated to hand hygiene and will not be used for cleaning medical devices.
By providing a clearly defined location for probe cleaning and high-level disinfection, this layout helps prevent workarounds, reduces cross-contamination risks associated with sink splash zones, and supports a consistent reprocessing workflow within the exam room.
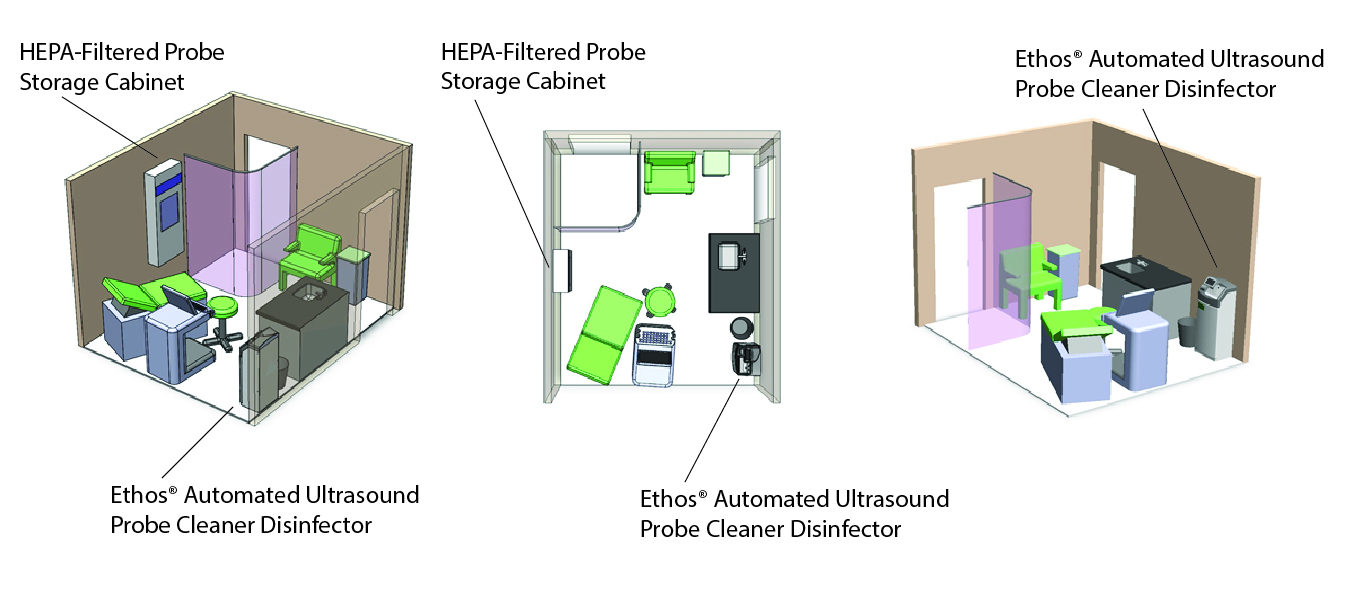
The placement of the Ethos in the above example room layout keeps probe reprocessing close to the point of care, minimizing unnecessary transport of contaminated probes through hallways while allowing staff to complete cleaning and high-level disinfection as part of the normal exam room workflow.
When exam rooms are designed intentionally, probe reprocessing does not need to compete with hand hygiene or patient care space. Instead, the equipment becomes a natural extension of the workflow.
If you have any infection prevention questions, contact Jill Holdsworth, Medical Affairs Manager.
