How to reduce the risk of HAIs
Ultrasound probes present special infection-prevention challenges. They are used across settings—from bedside point-of-care ultrasound (POCUS) to interventional suites and operating rooms. Because probes are used frequently, often on multiple patients in rapid succession, and may have complex housings or surface damage that harbors organisms, they can contribute to healthcare-associated infections (HAIs) if reprocessing and handling are not appropriate. Reducing probe-related HAIs requires clear risk-based categorization, strict adherence to manufacturer instructions and evidence-based guidelines, appropriate selection and use of covers and gels, validated cleaning and disinfection, staff competency, and systems that support safe workflows.
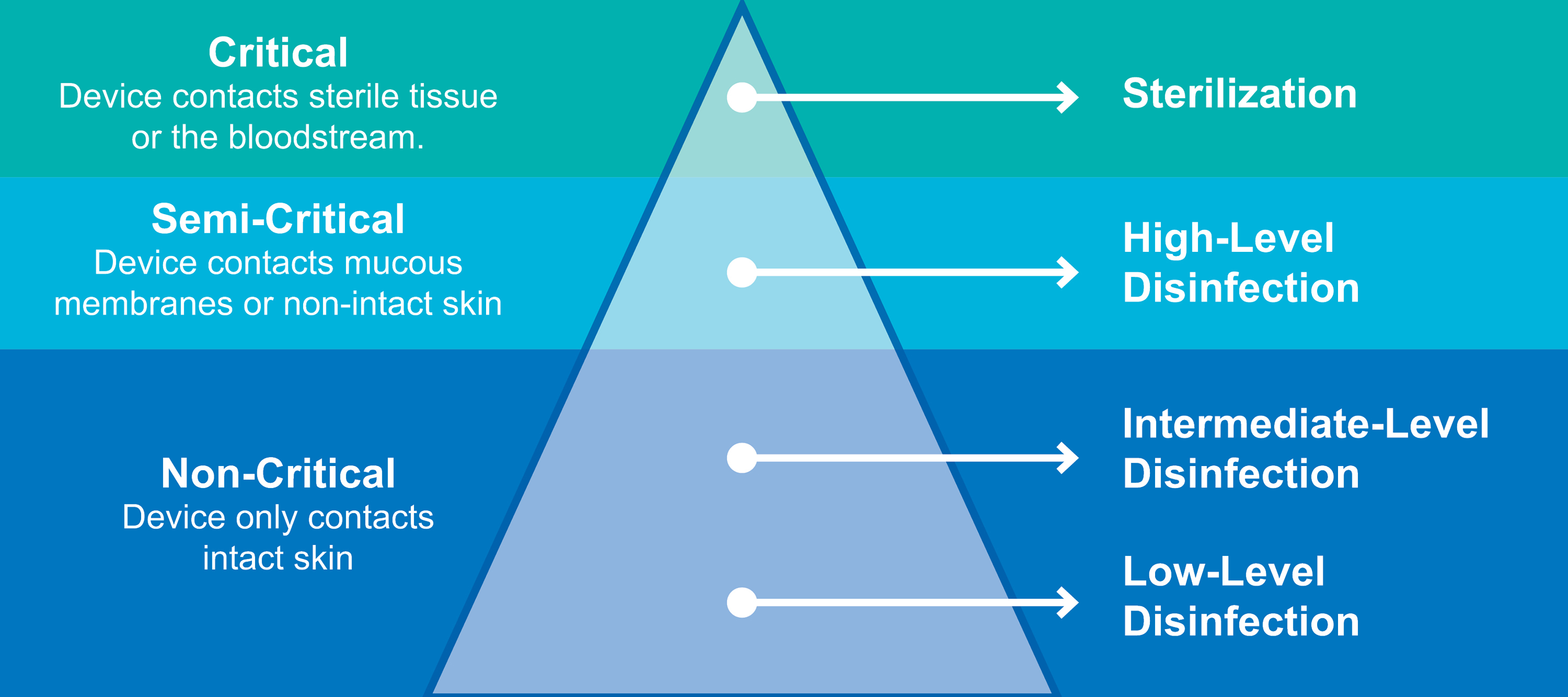
Risk categories and when higher-level reprocessing is needed
Non-critical use: Probes that contact intact skin only (e.g., many abdominal or vascular scans) are non-critical devices. Low-level disinfection (LLD) with an EPA-registered hospital disinfectant or detergent/disinfectant wipe is generally sufficient after each patient, combined with patient-considerate barrier practices (single-use gel packets, cleaning between patients).
Semi-critical use: Endocavity probes (vaginal and rectal) and any probe that contacts mucous membranes or non-intact skin require at minimum high-level disinfection (HLD) between patients. HLD kills vegetative bacteria, mycobacteria, fungi, and viruses (but not necessarily all spores) and is the recommended standard for these probes.
Point-of-use and cleaning fundamentals
- • Remove gel and gross contamination immediately after use. Leaving gel or organic material to dry markedly reduces the effectiveness of disinfectants and can promote biofilm formation.
- • Use probe and manufacturer-approved detergents and enzymatic cleaners. Cleaning, manual or automated, is the critical first step before any disinfection; it physically removes bioburden that could shield microbes from disinfectants.
- • Avoid immersing connectors, cable ends, or electrical interfaces unless explicitly allowed by the manufacturer. Many probes have seams or crevices; follow IFU for cleaning technique and any specialized brushes or tools recommended.
- • Inspect probes for damage (cracks, fissures, degraded membranes). Surface damage can harbor organisms and compromise the effectiveness of disinfection; damaged probes should be removed from service and repaired or replaced per policy to help reduce HAIs
Disinfection practices
For non-critical probes: Use low-level disinfection products per IFU and allow the required contact time. Single-use disinfectant wipes can be effective when used correctly.
For semi-critical probes (endocavity): Use high-level disinfection methods validated for the probe: cleaning agents and liquid HLD solutions (e.g., ortho-phthalaldehyde, glutaraldehyde, peracetic acid formulations) or automated systems designed for probes. Ensure correct concentration, temperature, contact time, and document cycles. Automated high-level disinfection systems can reduce human error when validated and maintained.
Rinsing and drying: After manual cleaning, follow IFU for rinsing (use sterile or properly filtered water if required) and thoroughly dry probes before moving from cleaning into HLD and then from HLD rinse to storage to prevent microbial proliferation and biofilm formation. Note: the incorporation of automated cleaning and HLD will eliminate the manual rinsing and drying step between cleaning and HLD as the device performs both tasks independently without separating the task. After drying, store probes in a HEPA-filtered storage cabinet to decrease the risk of HAIs.
Use of probe covers and gel
- • Probe covers (sheaths) are an adjunct, not a substitute for appropriate disinfection. Covers reduce contamination risk and preserve probe sterility during certain procedures, but they have documented failure rates (microscopic tears, slippage). Therefore, probes used with covers must still undergo cleaning and HLD when used in semi-critical applications.
- • Use sterile probe covers and sterile ultrasound gel for invasive or sterile-field procedures (e.g., biopsy, regional anesthesia) to minimize transfer of skin flora into the needle track or sterile site.
- • Avoid multi-use gel bottles at point of care; use single-use gel packets when possible and discard any opened multi-dose containers per facility policy.
Operational practices, training, and documentation
- • Develop and enforce written policies based on IFU and professional guidance (CDC, AAMI, APIC, HSPA, AIUM, specialty societies). When guideline recommendations differ, follow the device manufacturer’s IFU.
- • Train staff thoroughly on handling covers and gels, cleaning, disinfection steps, rinsing, and drying. Perform and document competency assessments regularly.
- • Implement process monitoring: logs of disinfection cycles, chemical indicator records, maintenance schedules, and periodic audits of cleaning technique.
- • Maintain traceability when possible: link probe use and reprocessing record to patient encounters to enable rapid investigation if an infection is suspected.

Special considerations for POCUS and high-throughput areas
- • POCUS probes are high-frequency, high-turnover devices in emergencies and critical-care settings. Establish realistic workflows that allow adequate cleaning and HLD where required—this may include having dedicated probes for invasive procedures, use of multiple probes to allow turnaround time, or access to automated reprocessor systems nearby to help reduce the risk of HAIs.
- • Avoid shortcuts such as skipping HLD after endocavity exams or relying solely on probe covers without subsequent disinfection.
Device procurement, maintenance, and innovations
- • When purchasing, prefer probes designed for easier cleaning and compatible with robust cleaning and disinfection options. Review automated probe reprocessors that can improve workflow and reduce handling that directly relates to probe damage. Consider disposable probe covers or single-use sterile sheaths.
- • Emerging technologies, such as automated probe reprocessors that clean and disinfect, may improve reliability and efficiency but must be validated against manufacturer guidance.
- • Proactive replacement of worn or damaged probes reduces risk from surface defects and biofilm niches.

Preventing probe-related HAIs requires a multi-layered, risk-based approach: classify probe use and apply the appropriate level of reprocessing; perform thorough cleaning, manual or automated, before any disinfection; use HLD for endocavity or mucous membrane-contacting probes; use sterile covers and sterile gel for invasive or sterile-field procedures; follow manufacturer IFUs exactly; train and assess staff competencies; and implement monitoring, documentation, and procurement strategies that prioritize patient safety. Organizational commitment—space, time, equipment, and policies that support correct reprocessing—is essential to translating best practices into consistent, safe care.
